
Stories
Ben Black: Risk, Opportunity, and Joy in Pushing the Bounds of Discovery

Ben Black attributes his early fascination with science to his fifth-grade teacher, a former industrial chemist, who lost a finger in a lab explosion. Black explains, “I can probably credit my teacher at the time, Al Nubling. He was just an incredible teacher—the best teacher I had in the public school. I didn’t have any scientists in my family, but Nubling had this background as an industrial chemist before he was a teacher, and he had sort of these ‘cool credentials’ because back in his chemist days, he had lost a finger in a lab explosion. That was quite exciting to hear about, and inspired this idea of, you know, mortal danger in the lab.”
But his dream crystallized on the day of the Challenger space shuttle disaster. Despite the tragedy, it was in that moment that Black decided that he wanted to pursue science as a career. He remembered that while his teacher was trying to be positive in explaining the accident to the class, Black could only think about being a biologist and working on a spaceship—there was something about the sense of danger and excitement associated with doing such innovative research that stuck out in his mind as a 10-year old. “I remember I decided that was what I was going to do, right then and there,” says Black.
By the time Black was in the ninth grade, he had decided that he would become a molecular biologist. He explains, “I had a biology teacher in high school, Hugh Nelson, and he initiated this program with a local biotech company, Amgen. The goal of the program was to bring recombinant DNA procedures and experimentation to high school labs.” Black was fortunate enough to participate in the early days of the innovative Amgen Biotech Experience science education program. There, he learned how to use recombinant DNA technology, attended lectures, and debated bioethical issues—all while still in high school.
Inspired by the engaging experience with the Amgen program, Black obtained a work permit and tried to apply for a job at biotech company—but to no avail. He continued to pursue entry-level laboratory positions, but his luck did not change for the better until he turned 18-years old. Black ultimately landed a job working at Novartis in agricultural biology, on a project to genetically modify maize. This was his first experience working in a lab full-time. Black recalls, “I really got interested in how chromosomes worked. I just knew I wanted to be a scientist in some capacity.”
Black feels thankful for all the help his mentors gave him—”Getting that positive push from the people you admire most can really keep you going for a while. Those experiences, those people—that’s where I actually became a scientist.”
At this time, Black pursued his undergraduate studies at Carleton College. There his pathway into science and discovery was guided by several academic advisers, including John Tymoczko and Stephan Zweifel. Tymoczko led an upper-level seminar series for students taught out of his living room. It was an engaging course that provided undergrads the experience of discussing foundational topics in biology with peers. Black also worked in the Zweifel lab where he was encouraged to keep up with published research articles in contemporary molecular biology in addition to completing lab work; this exposure helped prepare Black for graduate school. Reflecting on the value of a liberal-arts education, Black says, “There was encouragement to take classes in other disciplines outside of STEM and to push yourself to learn how to learn, get out of your comfort zone, and really deeply appreciate how much there is out there in the world that you don’t already know.”
After earning his bachelor’s degree, Black went to graduate school at the University of Virginia, where he worked in the lab of Bryce Paschal. Of that time, Black recalls, “I was in science heaven to carry out a Ph.D. in his group.” He remembers receiving a lot of encouragement from Paschal and others, and he ultimately had his choice of labs to join as a postdoctoral fellow. “I was lucky, I found myself able to go to Don Cleveland’s lab—at the University of California San Diego,” Black says. “He has the reputation as one of the top scientists in his entire generation in the biosciences.” Black notes that Cleveland exuded “this incredible enthusiasm, energy, and creativity; everybody was inspired that worked for him—including myself. I’d put it right up there as a career highlight even before I had my own lab and my own scientific independence.” Black feels thankful for all the help his mentors gave him—“Getting that positive push from the people you admire most can really keep you going for a while. Those experiences, those people—that’s where I actually became a scientist.”
“In grad school,” Black says, “I was thinking more broadly about how chromosomes function, how the cell nucleus functions to organize the DNA, and emerging concepts about how DNA-packing proteins—the so-called histones—could add epigenetic information on top of the DNA sequences.” Inspired by the work of his thesis committee chair, David Allis, on the histone code hypothesis—which states that modifications to histones impact downstream events such as gene expression—Black became more interested in the centromere, the part of the chromosome that directs inheritance through cell division and gamete formation. “So that was really a turning point, when I was deciding where the field was going, and thinking about how epigenetic information could be propagated through cell divisions and from generation to generation,” Black says. He took this passion with him to the University of Pennsylvania, where he was hired as an assistant professor and ultimately became a Rita Allen Foundation Scholar.
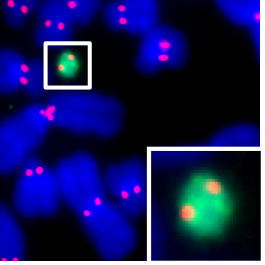
With support from the Rita Allen Foundation, Black and his team began work with connections to human health, disease treatments, and technological advancements in synthetic biology as it relates to the mammalian centromere. Black’s contributions have been in two areas: atomic-level structural and biophysical findings in the foundational centromere protein-DNA complexes and the discovery of biochemical pathways for centromere propagation. His lab made these advances, Black says, by “applying emerging DNA-sequencing technologies to provide the first evidence that centromeric DNA can function as a so-called selfish genetic element that is capable of driving non-Mendelian inheritance from one generation to the next.”
Recently, Black’s lab published two papers about using cryo-electron microscopy to generate a model of the core centromeric chromatin complex and build a new type of human artificial chromosome. “We think the new [artificial chromosomes] are a strong step forward to breaking through barriers that were holding back efforts to make synthetic mammalian genomes,” Black says. He is inspired by the work’s potential for the future—“We want to be part of the effort to take our own tools and push them further to help us understand chromosomes and to make synthetic chromosomes that will be useful for many, many potential applications in science and medicine. We’re really pumped about some of the recent progress and are thinking about the connections back to our original goals.”
So right away, it directly led to some of the most exciting work that’s come out of our lab and put my team on the map.
Black is now the Eldridge Reeves Johnson Foundation Professor of Biochemistry and Biophysics at UPENN. He shares reflections below on the impact of becoming a Rita Allen Foundation Scholar, changes in the scientific landscape, and advice for early-career scientists.
What do you remember about becoming a Rita Allen Foundation Scholar? How has that experience influenced the trajectory of your research?
I do think back to how it’s been important. On a practical level, it immediately accelerated our progress toward understanding how chromosomes are faithfully inherited through cell divisions. So right away, it directly led to some of the most exciting work that’s come out of our lab and put my team on the map. On a bigger level, it was maybe even more important to know that the Foundation supports creative work. For those of us trying to do something creative and important, when you’re starting your lab, there’s a bit of a Catch-22—there’s a lot of encouragement from folks to be creative, but then there’s also this pressure to secure funding for projects that are perhaps more conventional. The award gave us the funding to allow us to pursue some riskier things; these were ultimately more rewarding, and they were definitely just downright more fun to do.
What are your thoughts about the current research landscape?
If it wasn’t hard to do, it probably wouldn’t be worth the fight it takes to make new discoveries. I see it as an important challenge to continue to attract great minds and practitioners of science to career paths in the biosciences. We certainly don’t want to lose young, talented, creative, and passionate researchers. We should push ourselves to make sure that their individual paths in science are amply rewarded; that we’re encouraging risk-taking, creativity, and hard work.
What is your advice for early-career scientists pursuing this path?
Just be yourself. Know what you want in your research. Know from what in your research you derive joy, and then make it a priority to do that instead of what you think others want you to do. Reflecting on it and making sure you’re excited about what you’re doing—that’s always the most important thing, especially when you’re starting a lab and you have a lot of immediate challenges in front of you.
What gives you hope and optimism as you look ahead in your research?
Every step we take is another step toward an ultimate goal for the field; that’s what I’m most excited about right now. One of the things we’re thinking about is synthetic genome technologies. Now it’s bringing us to the point where we could really make some designer chromosomes. There’s the Human Genome Project-Write that Jef Boeke and George Church have initiated—that’s really exciting. They’ve been using synthetic-biology approaches coming from the J. Craig Venter Institute, including the synthetic prokaryotic organisms they’ve made. We’d like to connect in a more substantial way to those kinds of technologies and see where they can take us.